CU’s ClinImmune Labs enters private cord-blood banking arena
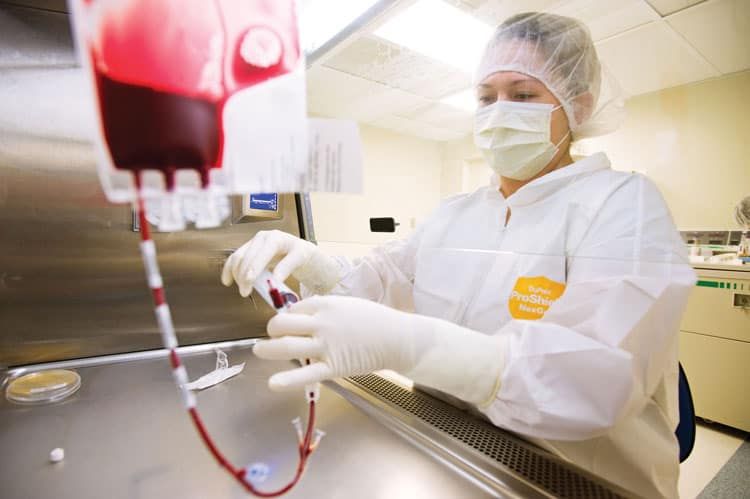

As the global market around umbilical cord blood stem-cell treatments reaches well into the billions of dollars annually, the University of Colorado has taken a pioneering step of sorts by expanding its own cord-blood banking services into the private realm.
ClinImmune Labs, a company owned by CU and housed at the school’s Anschutz Medical Campus in Aurora, has served as a public cord blood bank for 17 years. But ClinImmune last year began a public-private partnership with Irvine, Calif.-based CariCord Inc. to begin providing private banking.
The arrangement makes ClinImmune Labs the first Food and Drug Administration-licensed laboratory in the country doing private banking.
SPONSORED CONTENT
Select your Republic Services residential cart now!
In preparation for Republic Services becoming the primary provider of residential recycling, yard trimmings, and trash, residents should now select the best cart size and service schedule for their household needs.
Cord blood banking has grown in recent years as advances in medicine have increased the number of conditions and diseases that can be treated with transplanted cord-blood stem cells. That list includes more than 80 diseases, including certain types of leukemia and various blood, immune and metabolic disorders. Analysts for investment banking firm Jefferies estimated the 2010 global market for cord blood storage to be $4 billion.
The stem cells from cord blood are considered in some ways to be a better option than those from bone marrow. In addition to a lower chance of rejection by the recipient’s body, privately banked cord blood stem cells are also more readily available for children or their siblings to use, eliminating what can often be a months-long search for a bone marrow donor. Advocates of the practice have likened the cells to a “cellular legacy for the future.”
“The benefit of cord blood is it’s available immediately,” said Dr. Brian Freed, executive director of ClinImmune Labs and a co-founder of CariCord.
ClinImmune serves many functions in addition to cord-blood banking. Its services include stem cell processing, testing for bone marrow and organ transplants, and immunology testing, among others. The lab is owned, and supported in part, by the CU School of Medicine, but as a business it also generates nearly $10 million in revenue for the school each year.
Co-founded by Freed and company chief executive Calvin Cole, CariCord licenses the company name from CU and essentially works as the client-facing operation of the private banking now being done at the school. Parents banking their children’s blood pay CariCord, while CariCord contracts with the university for processing and banking.
“(CariCord doesn’t) have to worry about building the facility, and (ClinImmune Labs doesn’t) have to worry about advertising and sales and things we don’t know anything about,” Freed said.
CariCord charges $1,695 upfront plus about $125 each following year for banking. That’s similar to the other two major players in the industry, Cord Blood Registry and Viacord. CBR charges $1,650 upfront plus $150 per year and also offers a one-time lifetime fee of $4,000. Viacord charges $1,375 upfront and $175 per year.
Where CariCord’s advantage comes in, Freed said, is in the ClinImmune Labs bank, which has stored more than 8,500 donated cord blood units at its public bank, leading to more than 725 transplants at 150 different locations around the world. ClinImmune is the public banking site for Colorado parents who donate their babies’ cord blood, but it also has contracts with the states of Arizona and California to provide public banking services. No other private bank is based in Colorado.
While public cord-blood banks are required to gain FDA licensure, the requirements for private cord blood banks are much less stringent. In addition to skeptics questioning the value of private banking in relation to the cost, a 2014 Wall Street Journal report noted that the private banking industry has dealt with instances of “dirty storage conditions, leaky blood samples and firms going out of business.”
Such challenges make for an easy marketing pitch for CariCord and ClinImmune.
“Our attitude is, why not store them where we actually transplant them,” Freed said, adding that cord-blood transplants are done on a regular basis at CU. “You wouldn’t find that in any other private bank.”
Since launching CariCord in February 2014, Freed estimates that CariCord and ClinImmune have been doing about 50 private bankings per month.
Estimates vary as to where the cord-blood banking industry is headed. The Jefferies report estimated that the industry would grow from that $4 billion mark in 2010 to $15 billion this year.
Brian Tanquilut, a stock analyst at Jefferies, believes the industry’s growth has and will taper some because the global economic downturn impacted birth rates and made private banking too expensive for many families. But he acknowledges that the industry has no doubt grown rapidly.
Tanquilut said the competitive advantage a company such as CariCord might have in partnering with CU probably is negligible. Much of the consumer decisionmaking for cord banking in the United States, he said, is based on recommendations from physicians and the prices companies charge. If doctors start recommending CariCord because of ClinImmune Labs’ FDA licensure, though, he said that could make a difference.
“It’s a good branding tool,” Tanquilut said, “to be able to say, ‘We’re FDA approved,’ ”
Joshua Lindenstein can be reached at 303-630-1943, 970-416-7343 or jlindenstein@bizwestmedia.com. Follow him on Twitter at @joshlindenstein.

As the global market around umbilical cord blood stem-cell treatments reaches well into the billions of dollars annually, the University of Colorado has taken a pioneering step of sorts by expanding its own cord-blood banking services into the private realm.
ClinImmune Labs, a company owned by CU and housed at the school’s Anschutz Medical Campus in Aurora, has served as a public cord blood bank…
THIS ARTICLE IS FOR SUBSCRIBERS ONLY
Continue reading for less than $3 per week!
Get a month of award-winning local business news, trends and insights
Access award-winning content today!

